
Agile and Compliance in Medical Devices
(CT-IS103)
(CT-IS103)

Agile and Compliance in Medical Devices
(CT-IS103)
(CT-IS103)
This training is designed for medical device product development teams working under strict regulatory frameworks. Participants will learn how to align Agile practices with ISO 13485, ISO 14971, and FDA 21 CFR Part 820 compliance requirements. The course focuses on embedding traceability, validation, and documentation into Agile workflows without sacrificing speed or collaboration.
🎯 Learning Objective
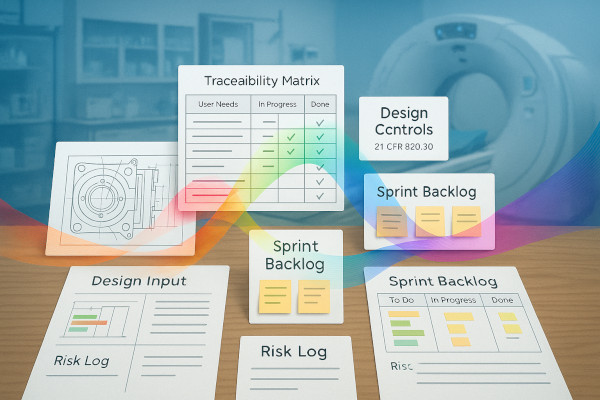
By the end of this training, participants will be able to:
- Map Agile artifacts (stories, backlogs, increments) to regulatory structures like DHF
- Ensure traceability across requirements, design, and verification
- Integrate design reviews and documentation checkpoints in Agile cadences
- Understand the compliance roles of Scrum Master, QA, and engineering leads
- Prepare for audits and regulatory submissions with Agile evidence
- Align sprint cycles with validation and release readiness criteria
📌 Who Should Attend
Level: Advanced
Team Format: Cross-functional teams preferred
🛠️ Format & Duration
🧭 Customization Options
Optional modules for IEC 62304 (software), MDR (EU), or local regulatory pathways.
🔗 Related Trainings
- CT-F105: Agile for Hardware and Systems Engineering
- CT-F109: Agile in Regulated Environments
- CT-M104: Agile Metrics and Reporting Essentials
